- Troposphere is the lowest level of the atmosphere, followed by the Thermosphere
- Good up High (Ozone is a protective gas in the Stratosphere)
- Bad Nearby (Pollutant in the Troposphere)
- Incomplete combustion results in the formation of Carbon Monoxide (CO)
- Catalytic converters catalyze the conversion of CO to C02 (reduces CO and also reduce VOC)
- Sulfur trioxide (SO3) forms corrosive sulfuric acid (H2S04) with water
- Sulfur dioxide reacts on the surface of a variety of airborne solid particles (aerosols), is soluble in water and can be oxidized within airborne water droplets, producing sulphuric acid.– (Ash hastens the formation of sulfur trioxide)
Category Archives: Research
TED
The Impact of Thalidomide On Society
Photo: Taken from http://www.wikiwand.com/de/Gr%C3%BCnenthal_(Unternehmen)
Origin of Thalidomide
Thalidomide was created by a family-owned pharmaceutical company, Chemie Grünenthal (Thalidomide Victims Association of Canada, 2017). Chemie Grünenthal was the first company to introduce Penicillin into the German market in the 1940s while in the 1960s, it introduced Thalidomide into the market.
Chemie Grünenthal first obtained a twenty-year patent for Thalidomide in 1954 and subsequently started clinical trials for the drug. Researchers at the company soon found out that the drug was rather effective in relieving morning sickness for pregnant mothers. Thalidomide was then marketed as Contergan® in the German market and was taken by many expectant mothers.
Beside West Germany where Thalidomide was first invented, Thalidomide was also distributed in the UK as Distaval, in Canada, and in the United States albeit without much success due to disapproval by the US Food and Drug Administration.
Impact of Thalidomide
Thalidomide was promoted as a ‘wonder drug’ to treat a range of conditions including headaches, insomnia and depression. It was popular because it was atoxic and so it was impossible to overdose on it (Thalidomide Society, N.A.). But long-term ingestion of the drug will result in irreversible peripheral neuritis, which is damage to the peripheral nerves, causing numbness, weakness and pain in the hands and feet (Mayo Clinic, 2017). The main impairments caused by thalidomide affect the limbs and are usually bilateral – either both arms or both legs or all four limbs (Thalidomide Society, N.A.).
Due to the absence of a census, there has not been an accurate number of fatalities resulted by Thalidomide. But according to the Thalidomide Victims Association of Canada, there were between ten and twenty thousand students born disabled as a result of the drug Thalidomide. The drug was responsible for deformities in children born by expectant mothers who consume it during their third trimester.
Impact of Thalidomide in Singapore
According to FFDN, the Thalidomide Association of Sweden , there is a personal anecdote of a lady born in the 1960s with disabilities in Singapore. She believed her defects at birth were a result of the consumption of Thalidomide by her mother during pregnancy. In her words, ‘there was an outbreak of infants born with deformed or missing limbs’. She received treatment as an infant which includes several operations and many hours of therapy to straighten her limbs. Growing up, she learnt how to walk with the help of callipers and walking frame.
Henceforth, against this personal narrative, there may perhaps be other children born with defects in the same period in Singapore which may have been left out of official records. Thus, Thalidomide did affect Singaporeans in the 1960s, although it is hard to estimate its impact.
Source: http://www.thalidomide.org/web/facts/
Chemistry Concept of Thalidomide
Researchers later realized that the problem lied in the fact that thalidomide was being provided as a mixture of two different isomeric forms.
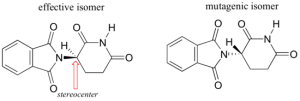
Diagram 1: Two isomers of Thalidomide
Both isomeric forms have the same molecular formula and the same atom-to-atom connectivity. Both isomers differ in the arrangement of the three-dimensional space about one tetrahedral, sp3-hybridized carbon. Hence, the two forms of thalidomide are known as stereoisomers. Tetrahedral carbons with four different substituent groups are called stereocenters as indicated in the diagram above




_logo.svg/440px-Gr%C3%BCnenthal_(Unternehmen)_logo.svg.png)