Question 1. In allergy sufferers, histamine causes runny noses, red eyes, and other symptoms. Here is its structural formula.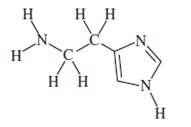
- Give the chemical formula for this compound.
- Circle the amine functional groups in histamine.
- Which part(s) of the molecule make the compound water soluble?
Answer:
- C5H9N3

- Amine functional group only.
Question 2. Antihistamines are widely used drugs for treating symptoms of allergies caused by reactions to histamine compounds. This class of drugs competes with histamine, occupying receptor sites on cells normally occupied by histamines. Here is the structure for a particular antihistamine.
- Give the chemical formula of this compound.
- What similarities do you see between this structure and that of histamine (shown in the previous question 1) that would allow the antihistamine to compete with histamine?
Answer:
- C16H21N3
- Pyridine
Question 3. Consider this statement. “Drugs can be broadly classed into two groups: those that produce a physiological response in the body and those that inhibit the growth of substances that cause infections.” Into with class does each of these drugs fall?
- aspirin
- morphine
- (Keflex) antibiotic
- estrogen
- amphetamine
- penicillin
Answer:
| Drugs that produce a physiological response in the body | Drugs that inhibit the growth of substances that cause infections |
| Aspirin, Estrogen, Morphine, Amphetamine | Penicillin, (Keflex) antibiotic |
Question 4. Herbal or alternative medicines are not regulated in the same way as prescription or OTC medicines. In particular, the issues of concern are identification and quantification of the active ingredient, quality control in manufacture, and side effects when the herbal remedy is used in conjunction with another alternative or prescription medicine.
- What do you think is the evidence from herbal supplement manufacturers that address these issues?
- Do you know anything about Singapore’s legislation on the topic?
Answer:
- Herbal supplement manufacturers may attain International Organisation for Standardization (ISO) Certification or the Certificate of Pharmaceutical Products (CPP) recommended by the World Health Organisation (WHO). Content of CPP should include the exporting (certifying) country, importing (requesting) country, recommended dosage and composition of products (active ingredients). Including the license holder details and date of issue of the product license can also address issues concerning the herbal medications.
- In Singapore, there is a Health Products Act in which manufacturers are required to provide information concerning its products and active ingredients. Manufacturers are also required to be licensed from Health Sciences Authority (HSA).

