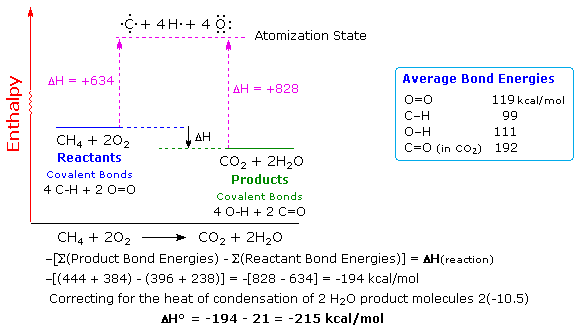
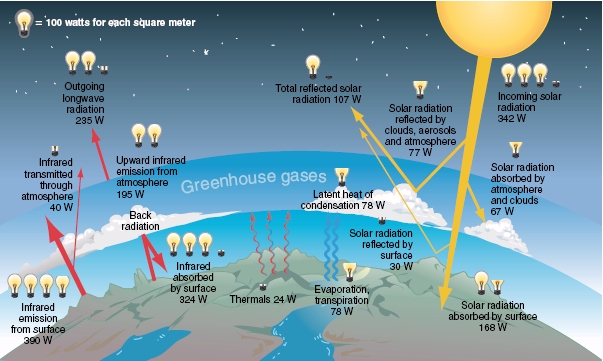
Q1: Understanding Earth’s energy balance is essential to understanding the issue of global warming. For example, the solar energy striking Earth’s surface averages 168 watts per square meter (W/m2), but the energy leaving Earth’s surface averages 390 W/m2. Why isn’t Earth cooling rapidly?
The amount of energy that Earth emitted (390W/m2) is more than twice the amount received (168W/m2). As the atmosphere absorbs quite a bit of the solar energy as heat energy, Earth does not cool rapidly (as seen from the yellow arrows in the picture below).
Q2: Decide and explain where the statement is correct or incorrect. “This winter has lowered my concerns about global warming…”. Explain.
The statement is incorrect because global warming will not cause an immediate effect on the four seasons we have. There is a change in climate due to global warming but not a change in weather.

The difference between climate and weather is that climate is the long term weather of a region, whereas weather is short term. In fact, due to global warming, we will have harsher winters due to the melting of artic ice. Hence, having winters does not improve the long term effects of global warming
Q3: One of the first radar devices developed during World War II used microwave radiation of a specific wave range that triggers the rotation of water molecules. Why was the design not successful?
The image below shows an example of a radar emission device used in World War I.

Since the wavelength of radiation used triggers the rotation of water molecules, it means that the radiation can also be absorbed by the atmospheric water vapour. When this radiation is absorbed by water vapour around the radar, it makes the radar unable to detect the target objects and doesn’t reach the target object.
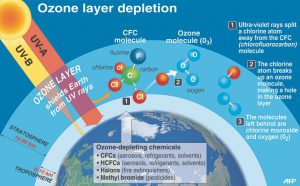
Q4: Now that you have studied air quality (Unit 1), stratospheric ozone depletion (Unit 2), and global warming (Unit 3),which do you believe poses the most serious problem for you in the short run (pick one and explain)? In the long run (pick one and explain)?
While the three problems are quite serious, they might affect us at different time intervals.
For example, air quality is a problem in the short run and can affect us on a daily basis, like the haze problem in Singapore. The stratospheric ozone depletion also affects us but is probably a major factor in the long run as the depletion process is relatively slow.

Lastly, global warming and climate change are influential in the long run as these effects are gradual and usually felt over long periods of time. Some future effects include increase in daily temperature, increased frequency of wamr days and nights, more intense and frequent rainfall, potential increase in wind speed and rise in sea level (as seen from the picture below).